METHYL OCTYL KETONE
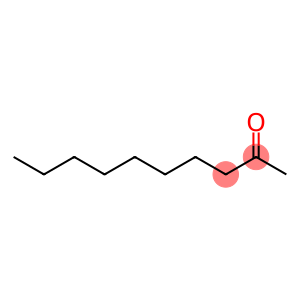
2-Decanone
CAS: 693-54-9
Molecular Formula: C10H20O
METHYL OCTYL KETONE - Names and Identifiers
| Name | 2-Decanone |
| Synonyms | DECANONE 2-Decanon 2-Decanone decan-2-one Decan-2-one n-C8H17COCH3 2-decanone+A8 octylmethylketone Octyl methyl ketone METHYL OCTYL KETONE Methyl n-octyl ketone METHYL N-OCTYL KETONE |
| CAS | 693-54-9 |
| EINECS | 211-752-6 |
| InChI | InChI=1/C10H20O/c1-3-4-5-6-7-8-9-10(2)11/h3-9H2,1-2H3 |
METHYL OCTYL KETONE - Physico-chemical Properties
| Molecular Formula | C10H20O |
| Molar Mass | 156.27 |
| Density | 0.825 g/mL at 25 °C (lit.) |
| Melting Point | 3.5 °C (lit.) |
| Boling Point | 211 °C (lit.) |
| Flash Point | 160°F |
| JECFA Number | 2074 |
| Water Solubility | Soluble in alcohol. Insoluble in water. |
| Vapor Presure | 0.248mmHg at 25°C |
| Vapor Density | >1 (vs air) |
| Appearance | neat |
| Color | Colorless to Almost colorless |
| BRN | 1747463 |
| Storage Condition | Store below +30°C. |
| Refractive Index | n20/D 1.425(lit.) |
METHYL OCTYL KETONE - Risk and Safety
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| UN IDs | NA 1993 / PGIII |
| WGK Germany | 2 |
| RTECS | HE0725000 |
| FLUKA BRAND F CODES | 10-23 |
| TSCA | Yes |
| HS Code | 29141990 |
| Toxicity | LD50 orl-mus: 7936 mg/kg TOLED5 30,13,86 |
METHYL OCTYL KETONE - Reference Information
| FEMA | 4271 | 2-DECANONE |
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| production method | 1. Tobacco: OR,26. 2. Preparation Method: In the reaction flask equipped with stirrer, ventilation tube, dropping funnel, add palladium chloride 0.53g(3mmol), cuprous chloride 2.97g(30mmol),24ml DMF aqueous solution (7:1), oxygen (bubbling) was introduced with stirring for about 1H. 1-Decene (2)4.2g(30mmol) was slowly added dropwise, and the addition was completed for about 10min. After the addition, oxygen was continued at room temperature. The reactant gradually changed from green to black, and after 15min, it turned green again. After aeration with stirring for 24h, the reaction mixture was poured into 100ml of cold 3mol/L hydrochloric acid and extracted five times with diethyl ether. The ether layers were combined, washed with saturated sodium bicarbonate solution and saturated brine successively, and dried over anhydrous magnesium sulfate. After rotary concentration, fractional distillation was performed under reduced pressure, and fractions of 43-50 ° C./133Pa were collected to obtain 3-3.4g (yield: 65%-73%) of acetodecone (1). [1] |
Last Update:2024-04-09 21:01:54
Supplier List
Multiple SpecificationsSpot supply
Product Name: Decan-2-one Visit Supplier Webpage Request for quotationCAS: 693-54-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Lactic Acid Impurity 40 Visit Supplier Webpage Request for quotationCAS: 693-54-9
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: 2-Decanone Request for quotation
CAS: 693-54-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 693-54-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2-Decanone Request for quotation
CAS: 693-54-9
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 693-54-9
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2-Decanone Visit Supplier Webpage Request for quotationCAS: 693-54-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: Decan-2-one Visit Supplier Webpage Request for quotationCAS: 693-54-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Lactic Acid Impurity 40 Visit Supplier Webpage Request for quotationCAS: 693-54-9
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: 2-Decanone Request for quotation
CAS: 693-54-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 693-54-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2-Decanone Request for quotation
CAS: 693-54-9
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 693-54-9
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2-Decanone Visit Supplier Webpage Request for quotationCAS: 693-54-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




